HTL-Strefa Inc. Responds To FDA Safety Alerts With European-Made Sicura™ Needles
In the wake of safety concerns flagged by the U.S. Food and Drug Administration (FDA) about certain plastic syringes and safety needles produced in China, HTL-Strefa Inc. steps forward with a robust solution. The company introduces DropSafe® Sicura™, a passive safety needle that sets new standards in healthcare safety and efficiency. This FDA-cleared product, crafted in Europe, is designed to meet the highest patient care and safety benchmarks.
Engineered in HTL-Strefa Inc.'s European facility, the DropSafe® Sicura™ passive safety needle is a testament to the company's dedication to quality. It addresses the critical need for safe and reliable medical supplies, especially in light of recent FDA evaluations. The needle features a transparent shield that locks automatically post-injection, ensuring a safer injection experience by significantly reducing the risk of needlestick injuries.
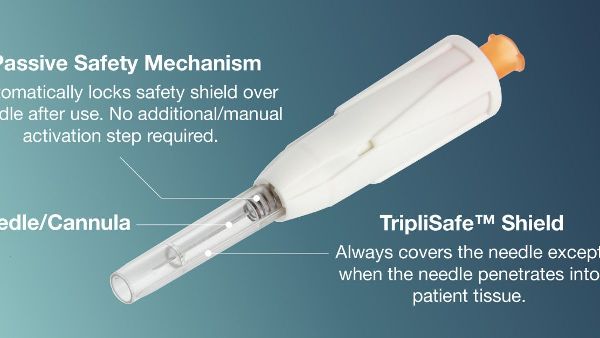
HTL-Strefa Inc. acknowledges the pressing need for dependable medical supplies amidst FDA's scrutiny over quality and performance issues of certain products. The introduction of DropSafe® Sicura™ passive safety needle not only underscores the company's commitment to safety but also assures its capability to fulfill growing demands without compromising on quality or performance. Carl Ward, General Manager at HTL-Strefa, Inc., highlighted the company's preparedness to support additional safety needle demand in North America, ensuring a smooth transition for healthcare systems towards safer alternatives.
With over three decades of service in the healthcare industry, HTL-Strefa Inc., along with its parent MTD Group, has been at the forefront of medical technology innovation. The company's dedication to improving clinical care safety and efficiency has led to the development of advanced technology, services, and solutions that support healthcare professionals and enhance patient outcomes.
For those interested in learning more about HTL-Strefa Inc.'s Sicura™ Passive Safety Needle and its commitment to quality and safety, further details can be found on their website or by contacting their customer support team.
About HTL-Strefa Inc. and MTD Group
HTL-Strefa Inc., together with MTD Group, stands as a leading figure in global medical technology. Specializing in the development, manufacture, and distribution of high-quality medical devices like passive safety needles, lancets, and pen needles, they are committed to innovation, safety, and customer satisfaction. Serving healthcare professionals and patients worldwide, their goal is to improve healthcare outcomes and enhance quality of life. For more information on HTL-Strefa Inc. and MTD Group or to connect with them on LinkedIn, please visit their respective websites.
Contact Information:
Emily Pupa
HTL-Strefa Inc.
113 Towne Lake Pkwy, Suite 120
Woodstock, GA 30188, USA
[email protected]
770-528-0410
This announcement from HTL-Strefa Inc. comes at a crucial time when the demand for safe and reliable medical supplies is at an all-time high. Their proactive approach not only addresses current challenges but also reassures healthcare providers and patients of their commitment to excellence in medical technology.
